Pages
6
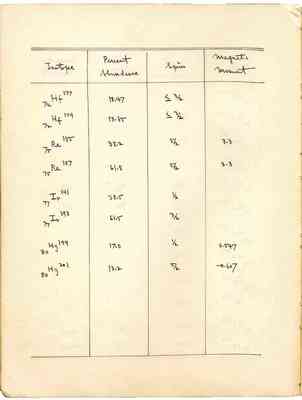
| Isotope | Percent Abundance | Spin | Magnetic Moment |
|---|
| [v]72[/v] Hf [^]177[/^] | 18.47 | less than or equal to 3/2 | |
|---|---|---|---|
| [v]72[/v] Hf [^]179[/^] | 13.85 | less than or equal to 3/2 | |
| [v]75[/v] Re [^]185[/^] | 38.2 | 5/2 | 3.3 |
| [v]75[/v] Re [^]187[/^] | 61.8 | 5/2 | 3.3 |
| [v]77[/v] Ir [^]191[/^] | 38.5 | 1/2 | |
| [v]77[/v] Ir [^]193[/^] | 61.5 | 3/2 | |
| [v]80[/v] Hg [^]199[/^] | 17.0 | 1/2 | 0.547 |
| [v]80[/v] Hg [^]201[/^] | 13.2 | 5/2 | - 0.607 |
7
August 2, '49
Neodymium NdCl3 Mol wt = 250.64 Sp. gr. = 4.134 Solubility 96.7 gms / 100 ml cold water 140 " / " " hot "
Samarium SmCl3 Mol. wt. = 256.80 Sp. gr. = 4.46 Solubility = 92.4 gms / 100 ml cold water 99.9 " / " " hot "
8
W. E. Lamb, Jr Phys. Rev. 60, 817, 1944
Diamagnetic correction to nuclear g factor g(1-0.319x10-4 z 4/3) using Thomas-Ferris field Using Hartree field, the coefficients are 19K , 20Ca , 26Fe , 29Cu , 37Rb , 55Cs , 74W , 80Hg 0.259 0.259 0.263 0.268 0.270 0.274 0.277 0.280
9
Compare two signals in the same magnetic field:
signal height recorded by the milliammeter
= C[j(j+1)γ^4 / (width)^2] (Molarity) (percent Abundance)
For Cd μCd = 0.65 j=1/2 NaCl 5.5 M
Na μNa = 2.215 j=3/2 CdCl2 2 [M]
(Signal/noise) ratio for Na observed in scope ~ [1₀?]
(Signal/noise) ratio for Cd recorded in meter ~0.0565 x 200 = 11.3 assuming 200 times enhanced.
10
August 6, 1949
Test of the size of the signal of na in 0.428 M NaCl soln plus 0.8 M MnSO₄. Hs = 0.8 gauss, Gain - 56, sweep freq = 91cps φ = 100
| No. of cells in the β-supply of oscillator | H₁ | Signal Ht | width |
|---|---|---|---|
| 1 | 86, 82, | 2 mm | |
| 1 1/2 | 30 | " | |
| 2 | 5 | " |
Using 0.2 M NaCl without adding Mn [bit?], under the same conditions as above (Hs = 0.8 gauss, gain - 56, H₁ corresponds to one cell) signal Ht = 4.5, 5.0, 4.5 width = (2mm)-
[Break]
Test: 0.1 M soln of NaCl was prepared by adding 0.2 M MnSO₄.