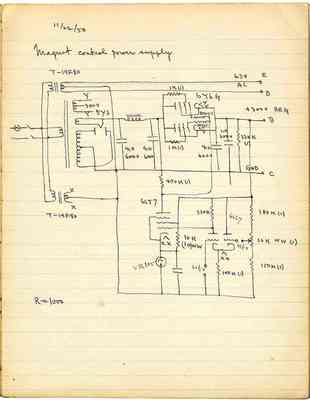
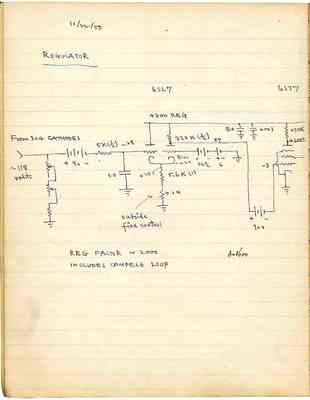
Pages
21
11/17/50
24.043 gms of triply distilled water was added to 4.225 gms MnSO₄·H₂O to form 25 cc. 1. Molar MnSO₄ solution
Percent H₂O in this solution = 97.9%
1/19/50
4.256 g. 99.5% D₂O was added to 0.507 g. MnSO₄·H₂O to form a 4 cc. solution.
Molarity of MnSO₄ = 3/4 Volume of D₂O - 3.830 cc Molarity of H = 1.28
24
This page is not transcribed, please help transcribe this page
25
11/23/50
Proton resonances from the following two samples were compared under identical conditions:
Sample volume = 4 cc Molarity of Mn++ = 0.75 Gain = 60 Hs = 0.086 G H₁ = 1 1/2 cell ~ 0.089 G Rate = D H₀ ~ 1500 G
(a) 4.256 g 99.5 % D₂O was added to 0.507 g. MnSO₄·H₂O to form 4 cc solution. It contains H to 1.28 M (b) 4 cc. of 0.75 M Mn++ in H₂O
Size of signal from sample (a) = 65.1
" " " " " (b) = 13.5 (attenuated to a factor 1/200)
Conclusion: [illegible] If attentuation factor is correct, % purity of D₂O is 98.2%