Pages That Need Review
Research Notes III, 1950
29
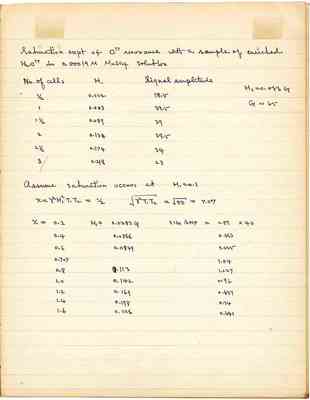
Saturation expt of O17 resonance with a sample of enriched H2O17 in 0.00019 M MnSO4 solution
| No. of cells | H₁ | Signal amplitude |
|---|---|---|
| 1/2 | 0.022 | 18.5 |
| 1 | 0.053 | 33.5 |
| 1 1/2 | 0.089 | 39 |
| 2 | 0.134 | 39.5 |
| 2 1/2 | 0.174 | 34 |
| 3 | 0.218 | 23 |
Assume saturation occurs at H₁ = 0.1
| x | H₁ | SIG AMP |
|---|---|---|
| 0.2 | 0.0283 G | 0.51 x 40 |
| 0.4 | 0.0566 | 0.864 |
| 0.6 | 0.0849 | 1.025 |
| 0.707 | - | 1.04 |
| 0.8 | 0.113 | 1.027 |
| 1.0 | 0.142 | 0.96 |
| 1.2 | 0.169 | 0.851 |
| 1.4 | 0.198 | 0.74 |
| 1.6 | 0.226 | 0.641 |
30
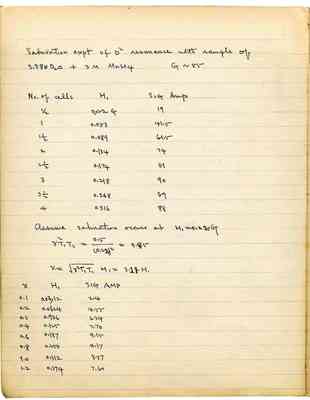
Saturation expt of D² resonance with sample of 3.38 N D₂O + 3 M MnSO₄
G ~ 55
| No. of cells | H₁ | Sig Amp |
|---|---|---|
| 1/2 | 0.022 G | 19 |
| 1 | 0.053 | 41.5 |
| 1 1 /2 | 0.089 | 61.5 |
| 2 | 0.134 | 74 |
| 2 1/2 | 0.174 | 81 |
| 3 | 0.218 | 90 |
| 3 1/2 | 0.268 | 89 |
| 4 | 0.316 | 88 |
| x | H₁ | SIG AMP |
|---|---|---|
| 0.1 | 0.0312 | 214 |
| 0.2 | 0.0624 | 4.55 |
| 0.3 | 0.936 | 6.34 |
| 0.4 | 0.125 | 7.70 |
| 0.6 | 0.187 | 9.15 |
| 0.8 | 0.250 | 9.17 |
| 1.0 | 0.312 | 8.57 |
| 1.2 | 0.374 | 7.60 |
31
12/10/50
The siqnal amplitudes & width of O17 & D2 resonances were compared at the same magnetic field
O17 Height = 41.7 ± 2.6 , Width = 40.0 ± 2.4 enriched H2O17 +0.00019M MnSO4
D2 1.20N D2O Height = 82 ± 4 Width = 66 ± 4 +3M MnSO4
a ≡ amplitude
I (I+1) n γ3 / ΔH2 ------------------------------------------------------------ ID(ID+1) nD γ3D / ΔH2D
= a ----- aD
I(I+1) = ID(ID+1)(nD / n)(γD / γ)3 {aΔH2 / aDΔH2}
= 2x 1.70/0.1 x 1.45 x 42x402/82x662 (1+14.5%) x 0.96 correction factor for difference in rates
=8.90 (1+14.5%)
I = 2.52 ± 0.[?] % abundance of O17 = 0.18 Molarity of O17 = 55.55 x 0.18 = 0.1
25
11/23/50
Proton resonances from the following two samples were compared under identical conditions:
Sample volume = 4 cc Molarity of Mn++ = 0.75 Gain = 60 Hs = 0.086 G H₁ = 1 1/2 cell ~ 0.089 G Rate = D H₀ ~ 1500 G
(a) 4.256 g 99.5 % D₂O was added to 0.507 g. MnSO₄·H₂O to form 4 cc solution. It contains H to 1.28 M (b) 4 cc. of 0.75 M Mn++ in H₂O
Size of signal from sample (a) = 65.1
" " " " " (b) = 13.5 (attenuated to a factor 1/200)
Conclusion: [illegible] If attentuation factor is correct, % purity of D₂O is 98.2%
10
Oct. 12, '50
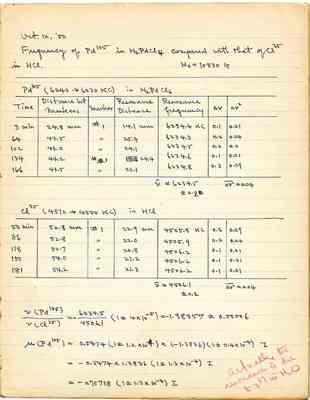
Frequency of Pd105 in H2PdCl4 compound with that of Cl35 in HCl.
H0 = 10830 G
[Break]
Pd105 (6240 --> 6230 KC) in H2PdCl6
| Time | Distance bet Markers | Marker | Resonance Distance | Resonance frequency | Δν | Δν² |
|---|---|---|---|---|---|---|
| 3 min | 24.8 mm | #1 | 14.1 mm | 6234.4 KC | 0.1 | 0.01 |
| 64 | 43.5 | " | 25.4 | 6234.3 | 0.2 | 0.04 |
| 102 | 43.0 | " | 24.1 | 6234.5 | 0.0 | 0.0 |
| 134 | 44.2 | # |
| 6234.6 | 0.1 | 0.01 |
| 166 | 43.5 | " | 23.1 | 6234.8 | 0.3 | 0.09 |
Cl35 (4510 --> 4500 KC) in HCl
| 50 min | 52.8 mm | 31 | 22.9 mm | 4505.8 KC | 0.3 | 0.09 |
|---|---|---|---|---|---|---|
| 86 | 52.8 | " | 20.8 | 4506.2 | 0.1 | 0.01 |
| 118 | 52.7 | " | 20.8 | 4506.2 | 0.1 | 0.01 |
| 181 | 54.2 | " | 21.3 | 4506.2 | 0.1 | 0.01 |
Actually the resonance is due to O17 in H2O
8
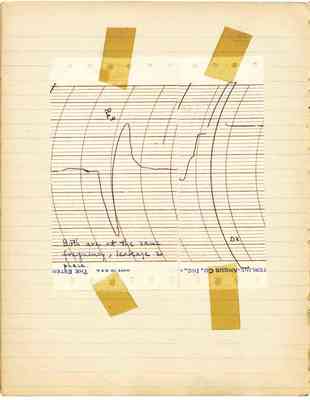
Be^9 Both are at the same frequency, leakage and phase. D^2
5
August 12, 1950 Preparation of Na3 Rh(NO2)6 m.w. [molecular weight] NaNO2 = 69.01 m.w. RhCl3 = 209.3 a.w. [atomic weight] Rh = 102.9
One gram of commercial RhCl3 was dissolved in 75 cc. of 1 m. NaNo2 & 5 cc. of 6 m. HC2H3O2; the mixture was heated at 60-90 degrees for 5 minutes. (A.A. Noyes & W. C. Bray, A sysem of qualitative analysis for rare elements, The MacMillan Company, 1948)
Sept. 2 - Sept. 12 Search for As, Zn, using KAsF6 & Zn(CH)4 + HF concentrated in water bath.
Zn 3.41 ML -> 3.015 ML at Ho = 11400 G