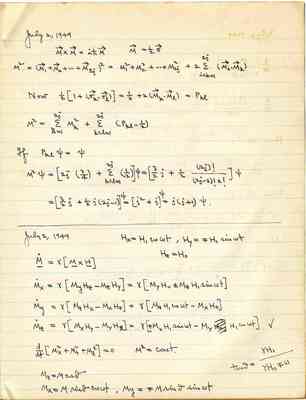Pages That Need Review
Research Notes I, 1949
70
Fine structure Expts
I. Fine structure of N14 resonance line was first noticed in sample containing liquid NH3 + NH4NO3 + Cr(NO3)2 · 9H2O
II. Demonstrated it was not Cr resonance by using samples
(1) Cr #1 12 M CrO3 in H2O (2) Cr #2 5M CrO2 in H2O + 1 M MnSO4 (3) Cr #3 1.25 M Cr(NO3)3
III. Crude expts show that Cu(NO3)2, Cr(NO3)2 and NH4NO3 give resonances nearer that freq of fine structure than nearer the big resonance
[diagram] [up arrow] Freq down
IV. Sample NH4NO3 + MnNO3 showed F.S. too !!!
75
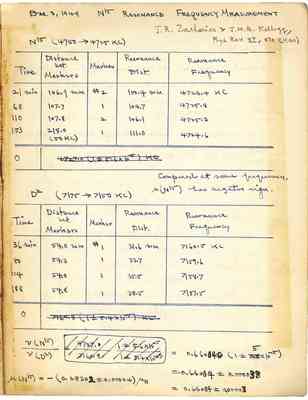
Dec 9,1949 N15 RESONANCE FREQUENCY MEASUREMENT J. R .Zacharias &J.M.B.Kellogg,Plus Rev. 57,570(1940) N15(4750->4725KC) Time Distance bet Markers Marker Resonance Dist Resonance Frequency 21Min 106.9mm #2 100.4mm 4726.4KC 68 107.7 1 104.7 4725.8 110 107.8 2 106.1 4725.3 153 218.0 1 111.0 4724.6 (50KC) Compared at same frequency, M(N15)has negative sign. D2 (7175->7150KC) Time Distance bet. Markers Marker Resonance Frequency 36min 54.0mm #1 31.6mm 7160.5KC 80 54.3 1 33.7 7159.6 124 54.0 1 35.5 7158.7 188 54.8 1 38.5 7157.5 0 V(N15)/V(D2)=4727.0/7160.8 1+-5.6x10-5/1+-5.4x10-5=0.660050(1+-5x10-5) =0.66004+-000033 =0.66004+-0.00003 M(N15)=-(0.283032+-0.00004)MN
77
H. Kopfermann [und] E. Rasmussen, Zeit f. Phys., 98, 624 (1936).
They were pretty sure that the spin of V51 is 7/2.
Auf Grunde dieser Überlegungen kommt zur Sicherstellung des Vanadium kernmoments wegen ihrer besonders großen J-Werte vor allem die Linie λ = 4591 Å 3d^3 4s^2 4F 9/2 - 3d^3 4s 4p ^4 G 11/2) in Frage. Es komiten die ersten fünf Komponenten und die Gesamtbreite ein wande frei gemessen werden. Eine schwache vorgelageite Komponente existiert nicht. Das Intervall verhältnis der gewesen Komponenten entspricht sehr genau dem Verhältnis der F-Werte das oberen Zustandes für I=7/2. Die daraus extrapotierte Gesamt breite stimmt mit der gemessen innenhalb der Fehlergrenzen überein. Der Wert I=9/20 für den zwar die Intervall regal auch noch genähert -- allerdings wesentlich schlecter als für I=7/2 -- erfüllt ist, scheidet mit Sicherheit aus, da ihm wegen der dann zu erwartenden zehn tashen Aufspaltung der Terme eine Gesamtbreite der Linie entsprechen würde, die um 0.05 cm-1 großer sein müßte, als die beobachtete Breite.
78
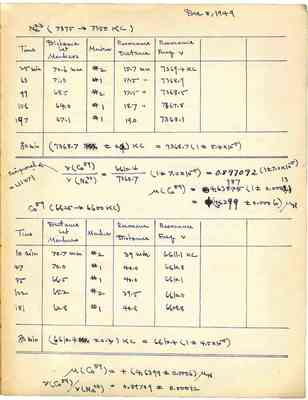
Dec 8, 1949 Na23 (7375 -> 7350 KC) Time Distance Markers Resonance Resonance bet Marker Distance Freq v 25 min 70.6 mm #2 15.7 mm 7369.4 KC 63 71.0 #1 17.5 " 7368.9 99 68.5 #2 17.5" 7368.5 156 64.0 #1 18.7" 7367.8 197 67.1 #1 19.0 7368.1 80 min (7368.7+-0.4)KC =7368.7(1+-5.4x10-5) Reciprocal=1.11471 v(Co59)/v(Na23)=6610.4/7368.7(1+-7.0x10-5)=0.897092(1+-7.0x10-5) M(Co59)+4.63987(1+-0.00013) =(4.6399+-0.0006)MN Co59(6625->6600KC) Time Distance bet Markers Marker Resonance Distance Resonance Freq v 10 min 70.7mm #2 39 min 6611.1 KC 47 70.0 #1 40.0 6610.8 85 66.5 #1 40.0 6610.1 122 65.2 #2 39.5 6610.0 181 62.8 #1 40.8 6608.8 80 min (6610.4+-0.3)KC=6610.4(1+-4.5x10-5) M(Co59)=+(4.6399+-0.0006)MN v(Co59)/v(Na23)=0.89709+-0.00012
79
Dec. 9 1949 Observations of the resonance of N14 were made with the following chemical compounds: K3 Fe(CN)6 , K4Fe(CN)6, K3 Co(CN)6. The results were N14 - resonance Compounds occures at same place K3Fe(CN)6 Fe has no 213 uB signal yes K4Fe(CN)6 yes K3Co(CN)6 This indicates that not only the complex K4Fe(CN)6 K3Co(CN)6 are diamagnetic, the Fe [illegible] Co in the compounds are themselves diamagnetic
83
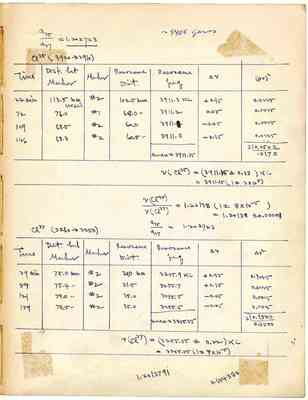
a35/a37 = 1.202723 ~ 9400 grams Cl35 (3920->3910) Time Dist. bet Marker Resonance [illegible] v [illegible]v2 Marker Dist. freq 22 min 113.5 mm (15 KC) #2 102.5 mm 3911.3 KC +0.15 0.0225 72 76.0 #1 68.0mm 3911.2 0.05 0.0025 109 68.5 #2 60.0 3911.1 -0.05 0.0025 146 68.3 #2 60.5mm 3911.0 -0.15 0.0225 mean=3911.15 3 0.0500 .0.170 v(cl35)=(3911.125+-0.13)KC =3911.15(1+-3x10 5) v(cl35)/v(cl37)=1.20138(1+-8x10-5) =1.20138+-0.00008 935/937=1.202723 Cl37(3260->3250) Time Dist bet Marker Marker Resonauce Dist Resonauce beg [?][?] 29min 75.0mm #2 30.0mm 3255.9KC +0.35 0.1225 84 75.4" #2 31.5 3255.7 +0.15 0.0225 121 79.0" #2 35.0 3255.5 -0.05 0.0025 159 78.5" #2 35.0 3255.5 -0.05 0.0025 mean=3255.55 3 0.1500 0.0500 v(cl37)=(3255.55+-0.22)KC =3255.55(1+-7x10-5) 1.2013791 0.684380
85
Dec 19, '49 Sample HCl #1 Cl35 Time=? #2 D2 3:40PM 4.56MC +5500 C 7.15MC+2360C 5450 2280 5450 2240 5450 2210 5450 2250 4.56546 MC 7.1523MC #3 Cl35 3:50 PM #4 D2 3:57 PM 4.56MC +4800 C 7.15MC +1330 4700 1230 4700 1280 4650 1220 4.5647 MC 7.1513 MC #5 Cl35 4:03 PM 4.56 MC+4300C at 3:50PM v(cl35)=4.5647 4300 v(D2)=7.1517 4300 4300 4.5643MC v(Cl35)/v(D2)=4.5647/7.1517=0.6382678 v(Cl35)/v(Na23)=4.5647/7.1517x[?]/0.26450+-0.01% 0.37043 Bitter's value=0.37051+-0.03% 0.37051+-0.00012
90
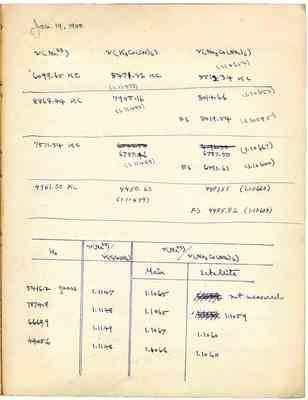
Jan. 19, 1950 v(Na23) v(K3Co(CN)6) v(Na3Co(NO2)6) (1.10654) 6099.65 KC 5471.86 KC 5512.34 KC (1.11473) 8868.44KC 7955.16 8014.66(1.10653) (1.11480) FS 8019.54(1.10585) 7511.54KC 6737.46 6787.50 (1.10667) (1.11489) FS 6791.61 (1.10600) 4961.50KC 4450.63 4483.55(1.10660) (1.11479) FS 4485.82(1.10604) Ho v(Na23)/v(K3Co(CN06) v(Na23)/v(Na3Co(no2)6) 5416.2 gauss 1.1147 1.1065 not measured 7874.8 1.1148 1.1065 1.1059 6669.9 1.1149 1.1067 1.1060 4405.6 1.1148 1.1066 1.1060